|
Therefore, the total number of protons and neutrons is called the atomic mass number. Therefore, atomic mass refers to the total mass of protons and neutrons.Ītomic mass (A) = nucleus mass = total mass of protons and neutrons (p + n)Īgain, the mass of each proton and neutron is about 1amu. The nucleus is made up of protons and neutrons. Therefore, the mass of the nucleus is called atomic mass. One is a positively charged particle proton and the other is a charge-neutral particle neutron.Īlmost all the mass of the atom is accumulated in the nucleus. There are two types of particles in the nucleus. We already know that the nucleus is at the center of the atom. Relationship between the atomic mass and sodium atomic number This site has published an article detailing the electron configuration of sodium that you can read if you want. To know these properties of sodium one must know the atomic number of sodium. The electron in the last shell of sodium easily forms a bond by donating to another element. Therefore, sodium is the s-block element. The last electron of sodium enters the s-orbital. So, the valence electrons of sodium are one. And the last shell has a total of an electron. The last shell of sodium has an unpaired electron, so the valency of sodium is 1. Therefore, the number of negatively charged electrons orbiting in its orbit is equal to the number of positively charged protons in the nucleus.Ītomic number (Z) = Number of charges in the nucleus (p) Importance of the atomic number of sodium That is the total number of protons in the atomic number. We know that protons are located in the nucleus of an atom as a positive charge. This number is equal to the serial number of the periodic table. The atomic number of the element is expressed by ‘Z’. Thus, the number of positive charges present in the nucleus of an element is called the atomic number of that element. He named that number the order of the atoms. The results of the experiment show that each element has a unique integer equal to the number of positive charges in the nucleus of that element. Scientist Henry Gwyn Jeffreys Moseley researched the X-ray spectrum of various elements in 1913-1914. Relationship between the atomic mass and sodium atomic number.Importance of the atomic number of sodium.Mårtensson, "Core-Level Binding Energies in Metals," J. 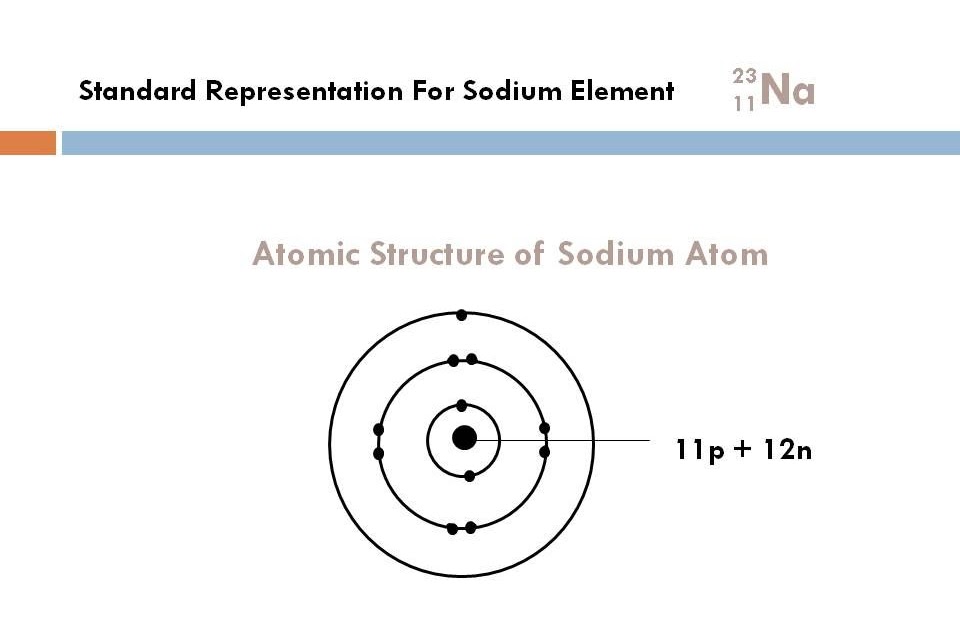
Lide, (Ed.) in Chemical Rubber Company handbook of chemistry and physics, CRC Press, Boca Raton, Florida, USA, 81st edition, 2000. Ley, Eds., Photoemission in Solids I: General Principles (Springer-Verlag, Berlin) with additional corrections, 1978. Burr, "Reevaluation of X-Ray Atomic Energy Levels," Rev. They are tabulated elsewhere on the WWW (reference 4) and in paper form (reference 5). The data are adapted from references 1-3. I am grateful to Gwyn Williams (Jefferson Laboratory, Virginia, USA) who provided the electron binding energy data. The binding energies are quoted relative to the vacuum level for rare gases and H 2, N 2, O 2, F 2, and Cl 2 molecules relative to the Fermi level for metals and relative to the top of the valence band for semiconductors. All values of electron binding energies are given in eV. 1967, 47, 1300.Įlectron binding energies Electron binding energies for sodium. These effective nuclear charges, Z eff, are adapted from the following references:
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |

 RSS Feed
RSS Feed
